Lesson
Introduction
Atomic structure is the foundation of chemistry, explaining the composition, properties and behaviour of matter. Every element and compound are made of atoms, and understanding how atoms are built helps us predict chemical reactions, bonding and periodic trends. In this blog, we explore the structure of the atom, subatomic particles, isotopes and electron configuration, along with examples, tables and comparisons to make learning clear and engaging.
This lesson is designed for students studying GCSE, IGCSE, CBSE, IB and the AP Curriculum, particularly those in Grades 9 to 11. It includes informative notes, interactive activity sheets, flashcards and engaging learning tasks to make this topic more enjoyable and easier to understand.
Components of an Atom
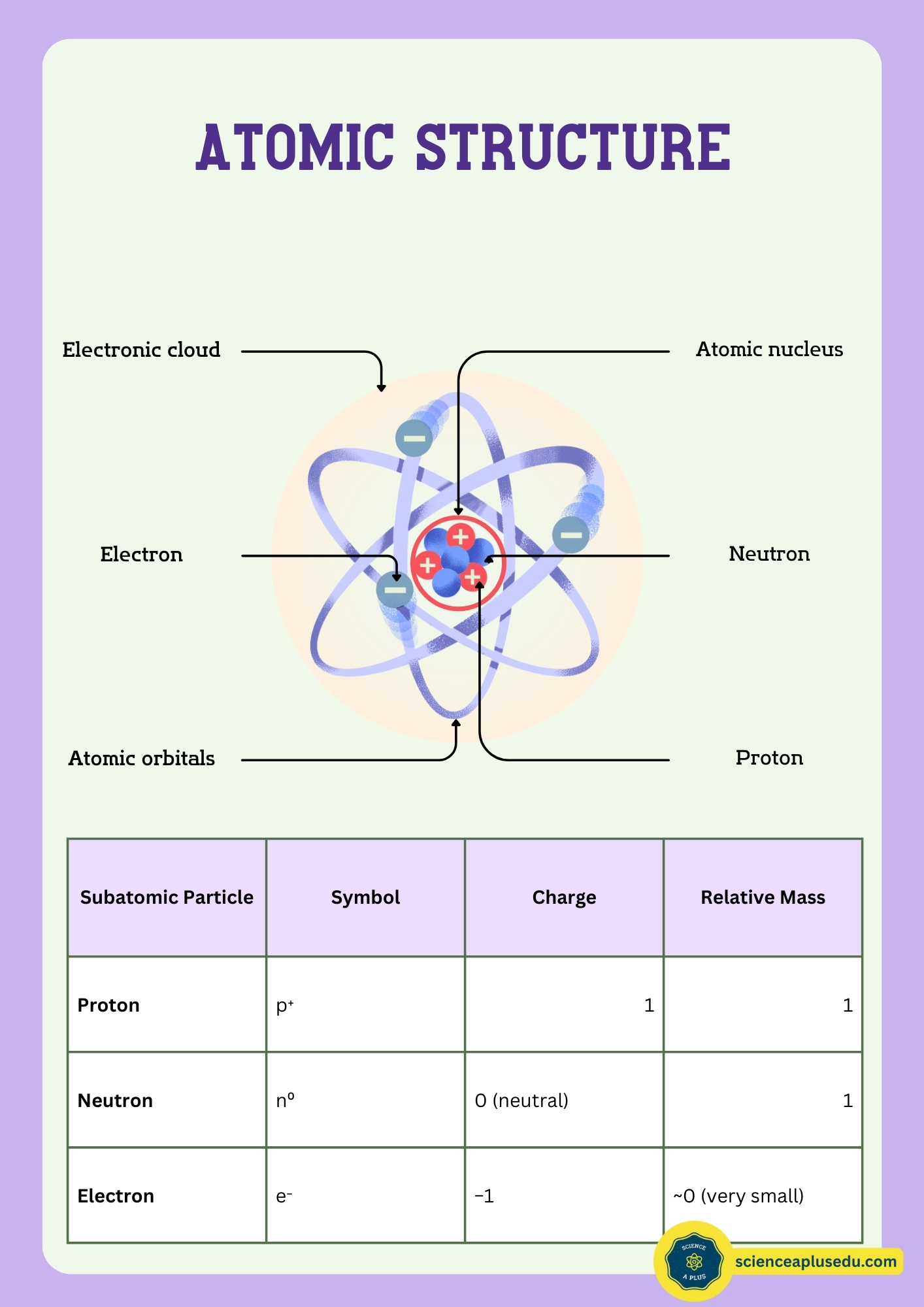
An atom consists of three primary subatomic particles:
Protons (p⁺)
- Positive charge (+1)
- Mass ≈ 1 u (atomic mass unit)
- Located in the nucleus
- Determines the atomic number and element identity
Neutrons (n⁰)
- Neutral (0 charge)
- Mass ≈ 1 u
- Located in the nucleus
- Contributes to mass number and isotope variations
Electrons (e⁻)
- Negative charge (-1)
- Mass ≈ 1/1836 u (very small)
- Orbit the nucleus in energy levels or shells
- Determines chemical reactivity and bonding
Atomic Structure and Subatomic Particles
Atomic Structure Diagram
Atomic Number and Mass Number
- Atomic number (Z): Number of protons in an atom
- Mass number (A): Sum of protons and neutrons
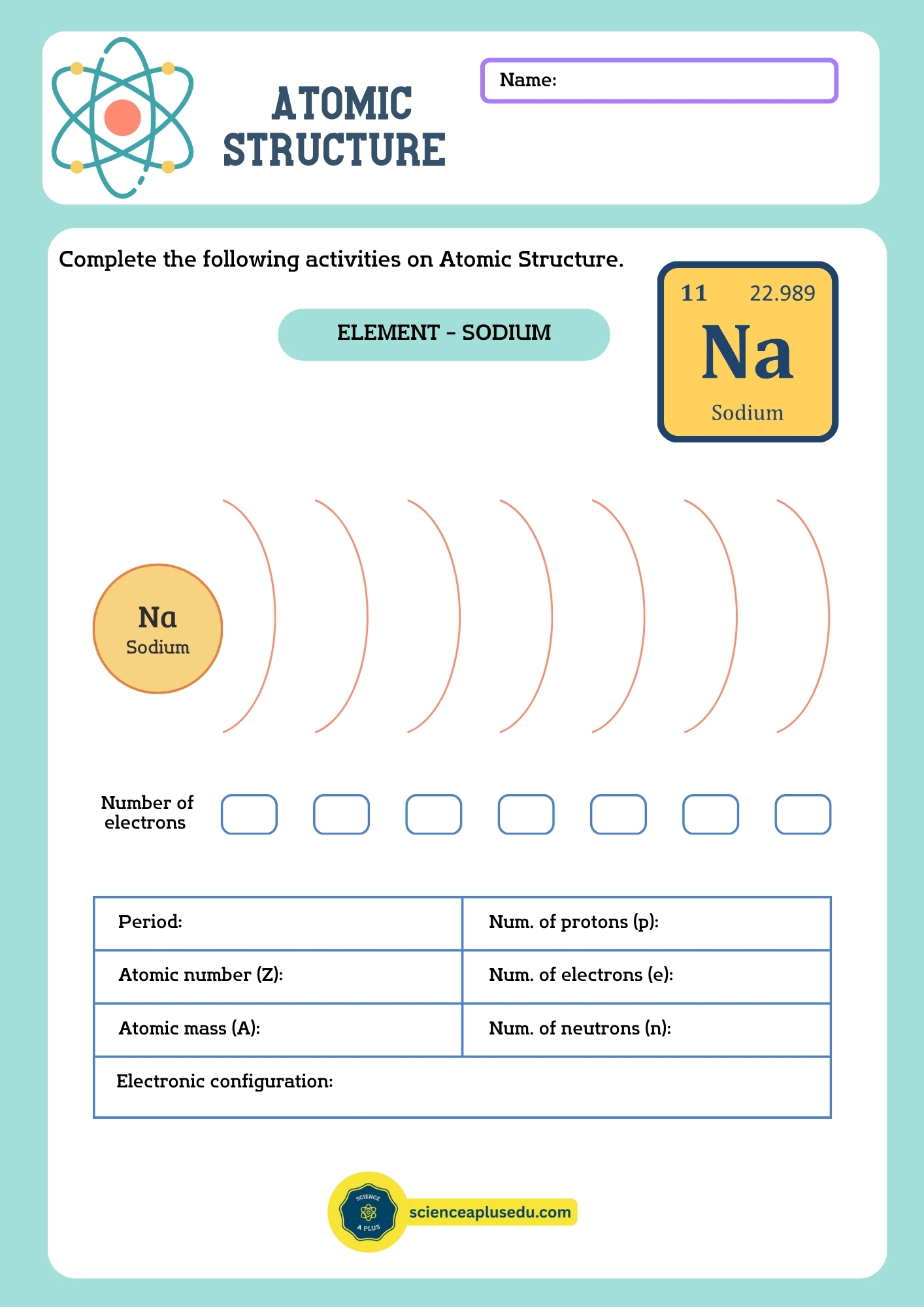
Example: Oxygen atom (O)
- Atomic number = 8 → 8 protons, 8 electrons
- Mass number = 16 → 16 – 8 = 8 neutrons

Isotopes
Isotopes are atoms of the same element with the same number of protons but different numbers of neutrons.
Example: Carbon
- Carbon-12: 6 protons, 6 neutrons
- Carbon-14: 6 protons, 8 neutrons
Applications of Isotopes:
- Carbon-14: Radiocarbon dating
- Iodine-131: Medical imaging
- Uranium-235: Nuclear fuel

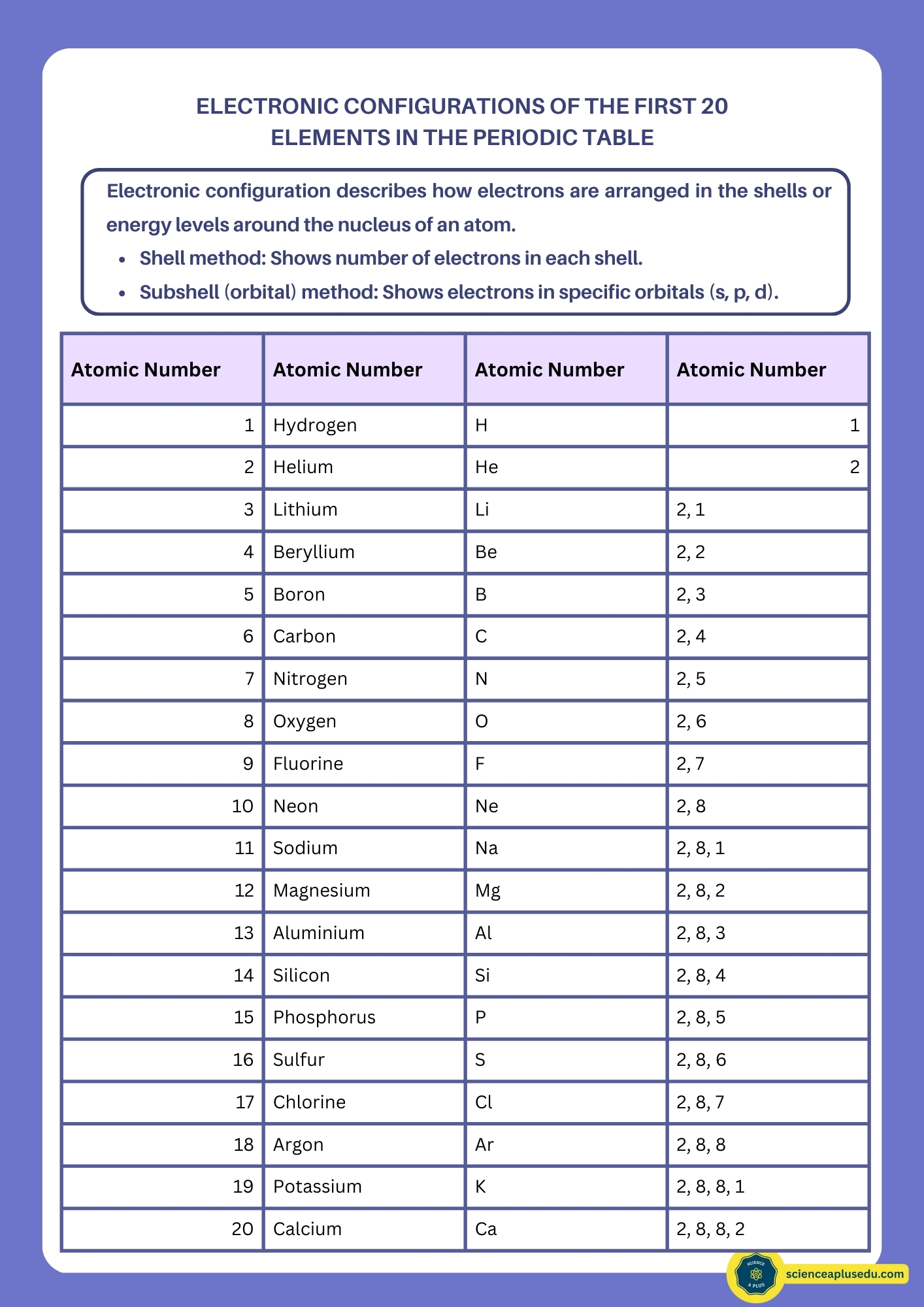
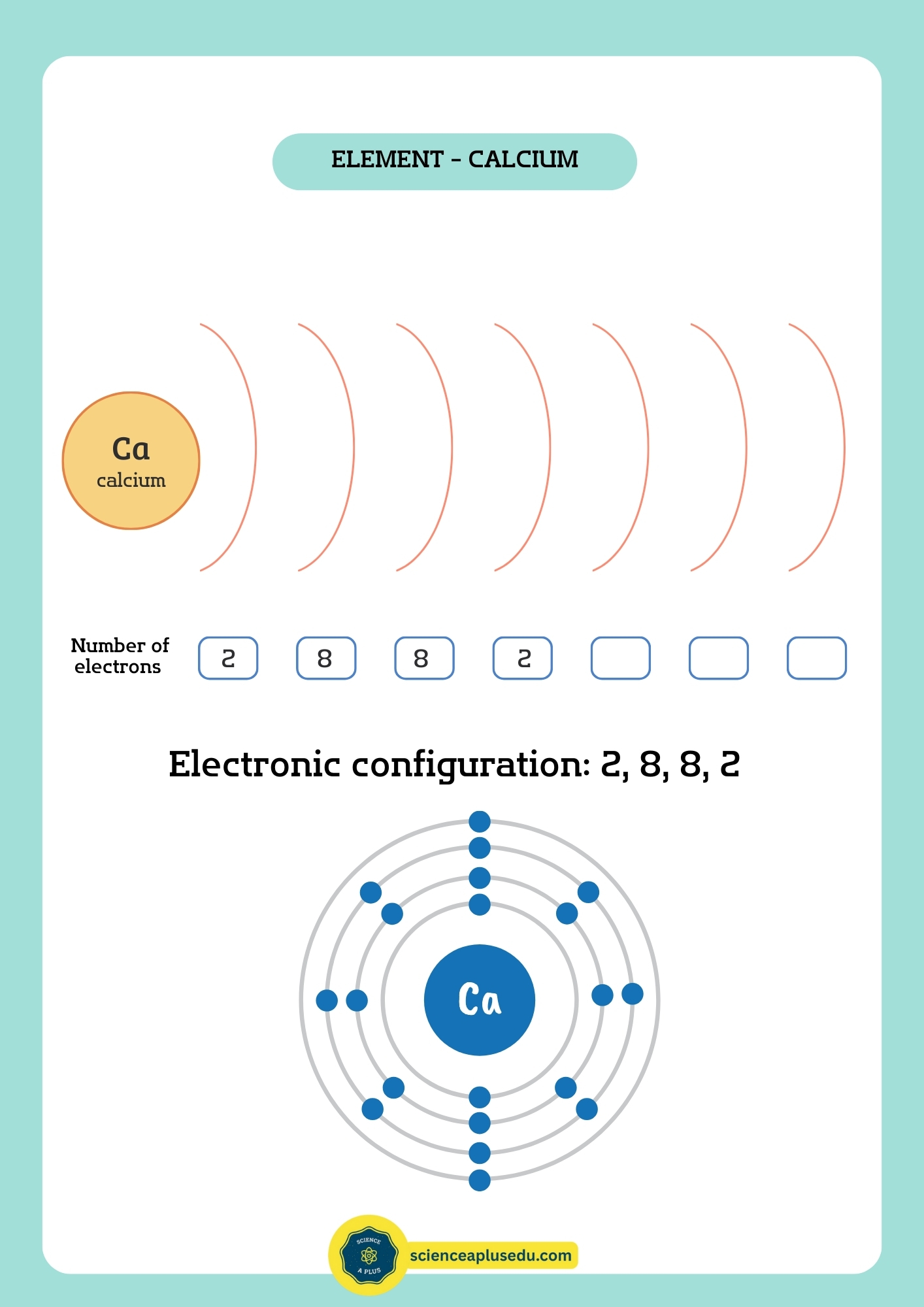
Electron Configuration
Electrons occupy energy levels (shells) around the nucleus: K, L, M, N, etc. Each shell has a maximum capacity:
- K: 2 electrons
- L: 8 electrons
- M: 18 electrons
- N: 32 electrons
Example: Oxygen (O), 8 electrons
- Shell K: 2 electrons
- Shell L: 6 electrons
- Configuration: 1s² 2s² 2p⁴
Element symbols and configurations
Electron Configuration - Calcium
Models of the Atom
- Dalton's Model (1803): Atoms are indivisible spheres.
- Thomson's Plum Pudding Model (1897): Atoms contain electrons in a positive sphere.
- Rutherford Model (1911): Small, dense nucleus; electrons orbit outside.
- Bohr Model (1913): Electrons occupy fixed energy levels.
- Quantum Mechanical Model: Electrons exist in clouds (orbitals); modern theory.
Applications of Atomic Structure
- Predicting chemical reactions
- Explaining periodic table trends
- Understanding bonding and molecular geometry
- Practical use in medicine, industry, and research
- Example: Hydrogen bonding in water explained by electron distribution
Conclusion
Atomic structure forms the foundation of chemistry, linking subatomic particles, isotopes, electron configuration, and chemical behaviour. Mastery of atomic structure helps students understand bonding, periodic trends, and reactions, providing the knowledge required for advanced studies in chemistry, physics, biology, and applied sciences.